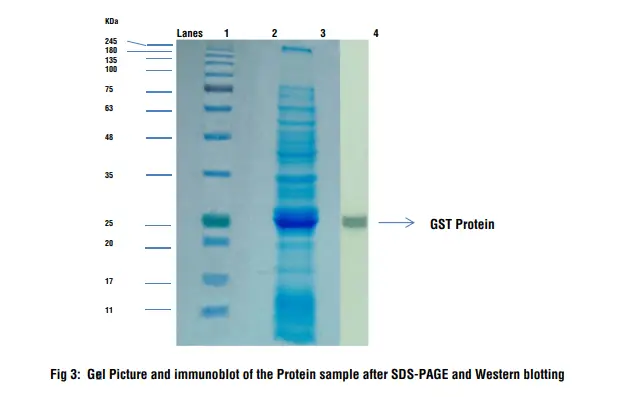
Used for protein sequencing, amino acid analysis, and solid phase assay systemsĪfter transfer, it is critical to block any remaining sites on the surface of the membrane to prevent the nonspecific binding of detection antibodies during subsequent steps.Generates higher background interference.Ideal for detecting high molecular weight proteins.Used for amino acid analysis and dot/slot blotting.Generates lower background interference.Ideal for detecting low molecular weight proteins.Physically durable and chemically resistant Proteins bind to membrane through hydrophobic and dipole interactionsīrittle, fragile and not chemically resistant Proteins bind to membrane through hydrophobic interactions 18000), or transferred onto a membrane for western blot analysis. Once separated by electrophoresis, proteins can be visualized in a gel using various stains, such as ProLite™ Orange (Cat No. In addition, SDS applies a negative charge equally across the length of the protein so that each molecule's movement through the gel, as driven by the electric field, is determined by its molecular weight only. The subsequent application of heat is used to remove the secondary and tertiary protein structure points so that each protein presents as one long linear macromolecule. In SDS-PAGE, the detergent sodium dodecyl sulfate is used to denature proteins and normalize their mass-to-charge ratios. There are several forms of PAGE, such as non-denaturing PAGE or native PAGE, which separate proteins based on their mass-to-charge ratios, but the most common is SDS-PAGE. Since proteins are complex macromolecules folded in on themselves, western blotting often uses a procedure such as PAGE (polyacrylamide gel electrophoresis) to provide information about its mass, charge, purity, and presence. PageTell™ protein standards can be conveniently loaded directly onto gels without any preparation (e.g., heat or additional loading buffer). Eleven of the proteins are covalently coupled with a blue chromophore except for two reference bands, a green and a red band at 25 kDa and 75 kDa, respectively, and when separated form distinctly visible bands. 6001) is a three-color protein standard that includes 13-prestained proteins ranging from 10 to 250 kDa in size. The PageTell™ Prestained 10 to 250 kDa Protein Ladder (Cat No. Standards include several proteins ranging in size from 10 to 250 kDa for comparison purposes and are available in various formats. For accurate size determination, a protein standard is run in conjunction with the sample. Gels typically have multiple channels or wells that separate several related experimental mixtures. Smaller proteins migrate faster than larger ones due to less resistance from the gel until pockets within the gel encapsulating proteins of similar size and charge are produced. In this method, an electric field forces proteins through the pores of a gel. Like many characterization techniques, western blotting uses gel electrophoresis to separate proteins based on size and charge. The CBB-stained gels were photographed using transmitted white light without an optical filter. The ProLite™ Orange stained gels were photographed using a SYPRO Orange filter. Three-fold dilution series of BSA standards were separated on a NuPAGE ® 4-12% Bis-Tris gel and stained with A) ProLite™ Orange Protein Gel Stain or B) Coomassie brilliant blue (CBB) according to standard protocols. 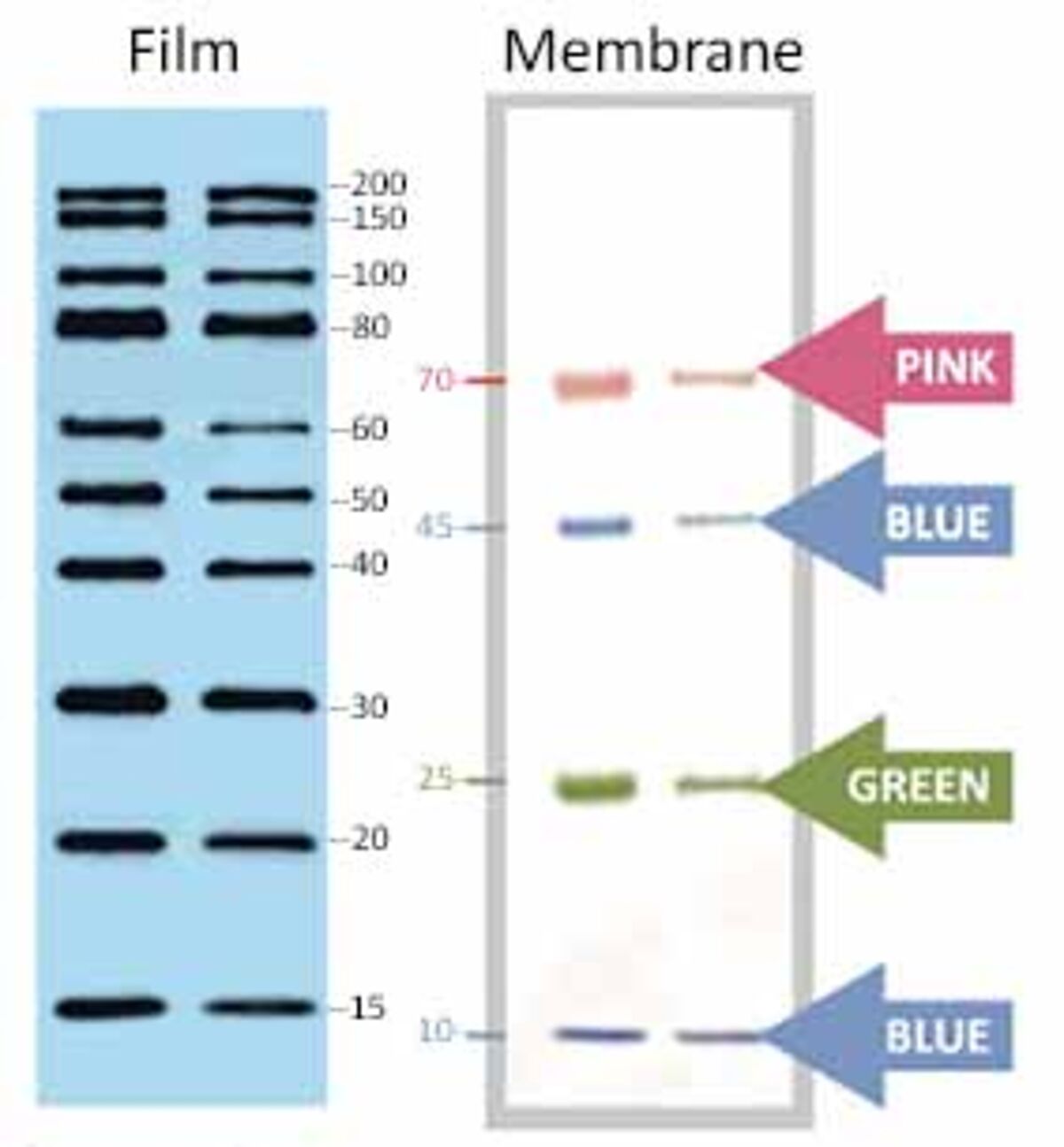
With the development of novel blotting technologies and specialized automated equipment, western blotting has become a cornerstone in proteomics research and protein detection, producing qualitative and semi-quantitative results. Western blotting is used extensively in biochemistry applications targeting complex proteins, testing for disease markers (e.g., HIV, Lyme disease, or Hepatitis B), and confirming protein production in cloning experiments. Common reporter molecules include enzymes, such as horseradish peroxidase (HRP) and alkaline phosphatase (ALP), or fluorescent dyes, such as iFluor® 488, with the latter affording greater sensitivity and multiplexing capacity. Antibodies are selected based on their specificity for the protein of interest, and this specificity of the antibody-protein interaction enables target identification. This process involves the electrophoretic separation of proteins, the transfer of separated proteins from a gel to a stable membrane substrate, and their subsequent detection by antibodies labeled with reporter molecules. Western blotting is an analytical technique used to detect the presence of specific proteins in a complex biological sample.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
